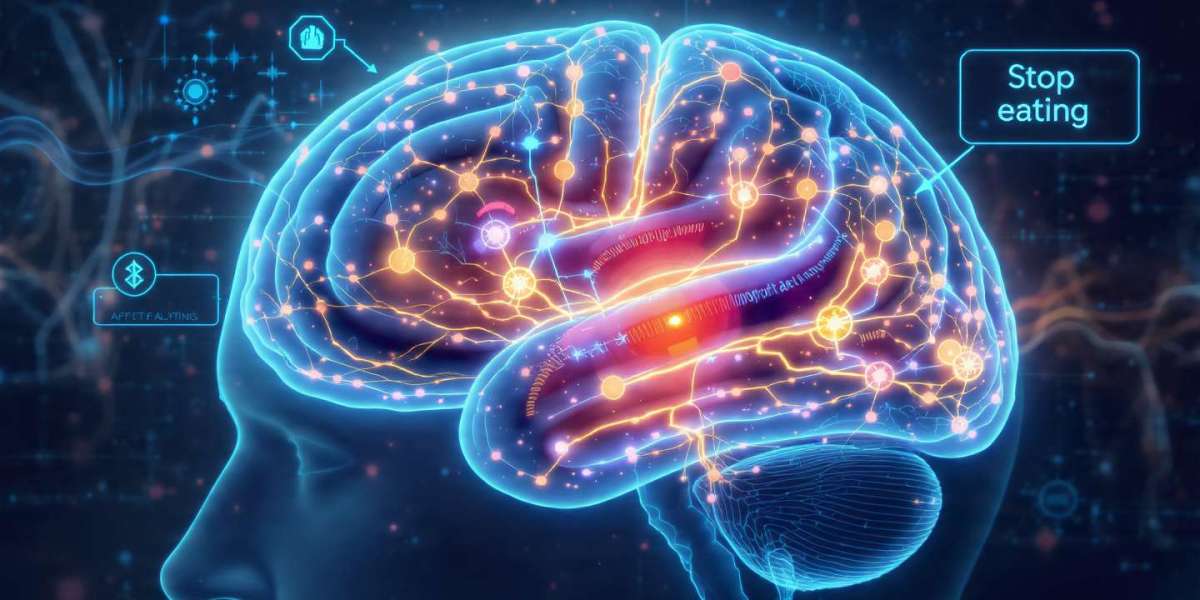
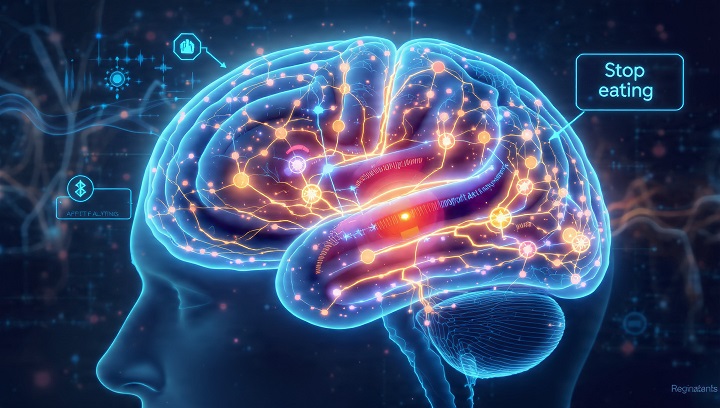
Scientists Discover Hidden Brain “Stop Eating” Switch: How Astrocytes Control Appetite
Key Points:
Astrocytes, once thought to be support cells, actively signal fullness to neurons.
Tanycytes detect glucose and trigger a chain reaction leading to appetite suppression.
This discovery could open new treatments for obesity and eating disorders.
advertisement
Introduction
For decades, scientists believed neurons were the sole masterminds behind hunger and fullness. Recent groundbreaking research from the University of Maryland, in collaboration with the University of Concepción in Chile, challenges this notion. Scientists have uncovered a previously unknown brain communication pathway, revealing that astrocytes—cells long considered mere neuronal support—play a crucial role in telling the brain when to stop eating.
This discovery not only reshapes our understanding of appetite control but also hints at innovative approaches for treating obesity and eating disorders.
How the Brain Detects Fullness: The Role of Tanycytes
illustrative image
The process begins with tanycytes, specialized cells lining a fluid-filled cavity deep in the brain. After a meal, rising glucose levels in cerebrospinal fluid are detected by these cells. Tanycytes then metabolize the sugar and release lactate, a metabolic byproduct, into surrounding brain tissue.
Previously, researchers assumed that lactate acted directly on neurons. However, the new study shows that astrocytes serve as a critical middleman in this signaling pathway, fundamentally altering our understanding of appetite regulation.
Astrocytes: The Unexpected Messengers
illustrative image
Astrocytes, one of the most abundant brain cell types, were historically viewed as “support staff” for neurons. The study, published on April 6, 2026, in the Proceedings of the National Academy of Sciences, revealed that astrocytes carry HCAR1 receptors, which detect lactate from tanycytes. When activated, astrocytes release glutamate, a chemical messenger that communicates directly with appetite-suppressing neurons, producing the sensation of fullness.
Professor Ricardo Araneda from University of Maryland explains: “Tanycytes ‘talk’ to astrocytes, and then astrocytes ‘talk’ to neurons. This complex conversation fundamentally changes how we think about brain circuits controlling eating.”
A Chain Reaction Across the Brain
illustrative image
Experiments demonstrated that stimulating a single tanycyte could trigger activity in multiple surrounding astrocytes, highlighting the expansive network effect. Interestingly, lactate may have a dual impact: activating fullness neurons via astrocytes while potentially suppressing hunger-promoting neurons through a separate, direct route.
This dual mechanism offers exciting possibilities for precision interventions in appetite regulation, potentially benefiting individuals struggling with overeating or irregular eating patterns.
Implications for Obesity and Eating Disorders
illustrative image
Although the current research was conducted in animal models, both tanycytes and astrocytes are present in all mammals, including humans. This suggests that the same signaling mechanism could operate in people.
The next steps for researchers include exploring whether manipulating the HCAR1 receptor in astrocytes can influence eating behavior. While no drugs yet target this pathway directly, the discovery points toward a new therapeutic direction that could complement existing obesity treatments like Ozempic.
A Decade of Collaborative Science

This study represents nearly ten years of collaboration between Araneda’s lab at University of Maryland and María de los Ángeles García-Robles’s team at the University of Concepción. The lead author, doctoral student Sergio López, conducted pivotal experiments during an eight-month research visit to Maryland. The work was funded by Chile’s National Fund for Scientific and Technological Development, the Millennium Institute of Neuroscience, and the U.S. National Institutes of Health.
Conclusion
The discovery of astrocytes as active regulators of appetite revolutionizes how we understand brain signaling. By revealing the hidden “stop eating” switch, this research opens new pathways for tackling obesity, eating disorders, and other appetite-related conditions. Beyond clinical implications, the study highlights the beauty and complexity of the brain’s internal communication network—reminding us that even the smallest cellular interactions can profoundly influence our behavior and health.
As scientists continue exploring this hidden signaling chain, the future may hold targeted therapies that help millions maintain healthier eating habits, combining innovation with an unprecedented understanding of the brain’s inner workings.
Key Points Summary
Astrocytes actively regulate appetite by signaling neurons when we are full.
Tanycytes detect glucose after eating and release lactate to activate astrocytes.
Discovery could lead to new therapies for obesity and eating disorders.
Research conducted by University of Maryland and University of Concepción.
Pathway involves HCAR1 receptor and glutamate signaling in the hypothalamus.
advertisement
Frequently Asked Questions (FAQ)
Q1: What are astrocytes?
Astrocytes are a type of brain cell traditionally considered support cells for neurons but now found to actively regulate appetite.
Q2: What role do tanycytes play in hunger control?
Tanycytes detect glucose after meals and release lactate, which activates astrocytes to signal fullness.
Q3: Could this discovery help humans?
Yes. Since tanycytes and astrocytes exist in all mammals, the mechanism likely exists in humans, offering potential new treatments for obesity and eating disorders.
Q4: Are there drugs that target this pathway?
Currently, no drugs directly target the astrocyte HCAR1 pathway, but it may become a promising target alongside existing treatments like Ozempic.
Q5: How long did the research take?
Nearly a decade of collaborative work between the University of Maryland and University of Concepción.
Sources
ScienceDaily – Discovery of astrocytes’ role in appetite regulation
https://www.sciencedaily.com/releases/2026/04/260406192811.htmEurekAlert – Hidden brain switch controlling hunger identified
https://www.eurekalert.org/news-releases/1122997
Thank you !