article image source: nature.com (Link)
Scientists Create Powerful New Aluminum Material That Could Replace Rare Earth Metals
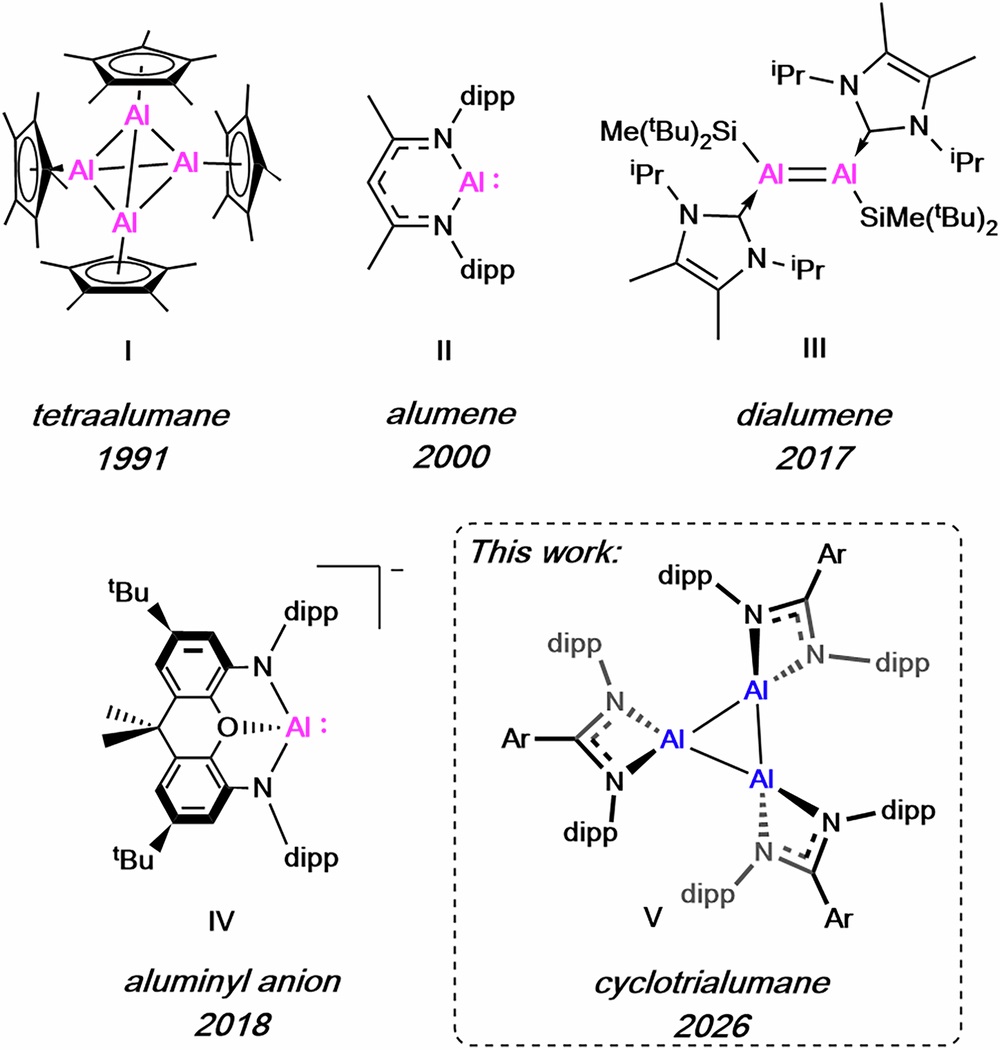
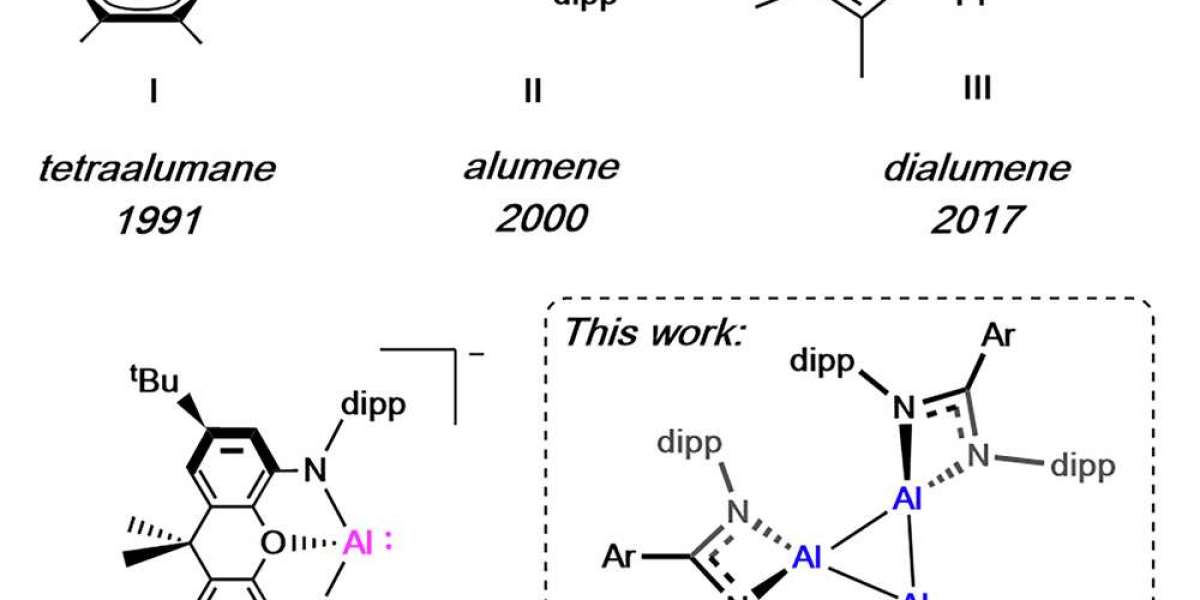
I Tetraalumane, Schnöckel, 1991; II Alumene, Roesky, 2000; III Dialumene, Inoue, 2017; IV Aluminyl anion, Aldridge and Goicoechea, 2018; V This work: Cyclotrialumane.
image source: nature.com
- Scientists have discovered a new molecular form of aluminum capable of breaking extremely strong chemical bonds.
- The breakthrough could reduce reliance on rare earth and precious metals like platinum and palladium.
- Researchers say the discovery may enable cheaper, greener, and more sustainable chemical manufacturing.
advertisement
A New Form of Aluminum Could Transform Modern Industry
Scientists have developed a powerful new form of aluminum that could replace rare earth metals, potentially transforming the future of chemical manufacturing and advanced technologies.
Researchers from King’s College London recently discovered an unusual aluminum structure with chemical properties that challenge long-standing assumptions about how this common metal behaves.
The discovery suggests that aluminum — one of the most abundant metals in Earth’s crust — may become a much cheaper and more sustainable alternative to rare and expensive metals widely used in modern industry.
Their findings were published in Nature Communications, highlighting a breakthrough that could reshape how chemists design materials and perform complex chemical reactions.
A Rare Molecular Structure Never Seen Before
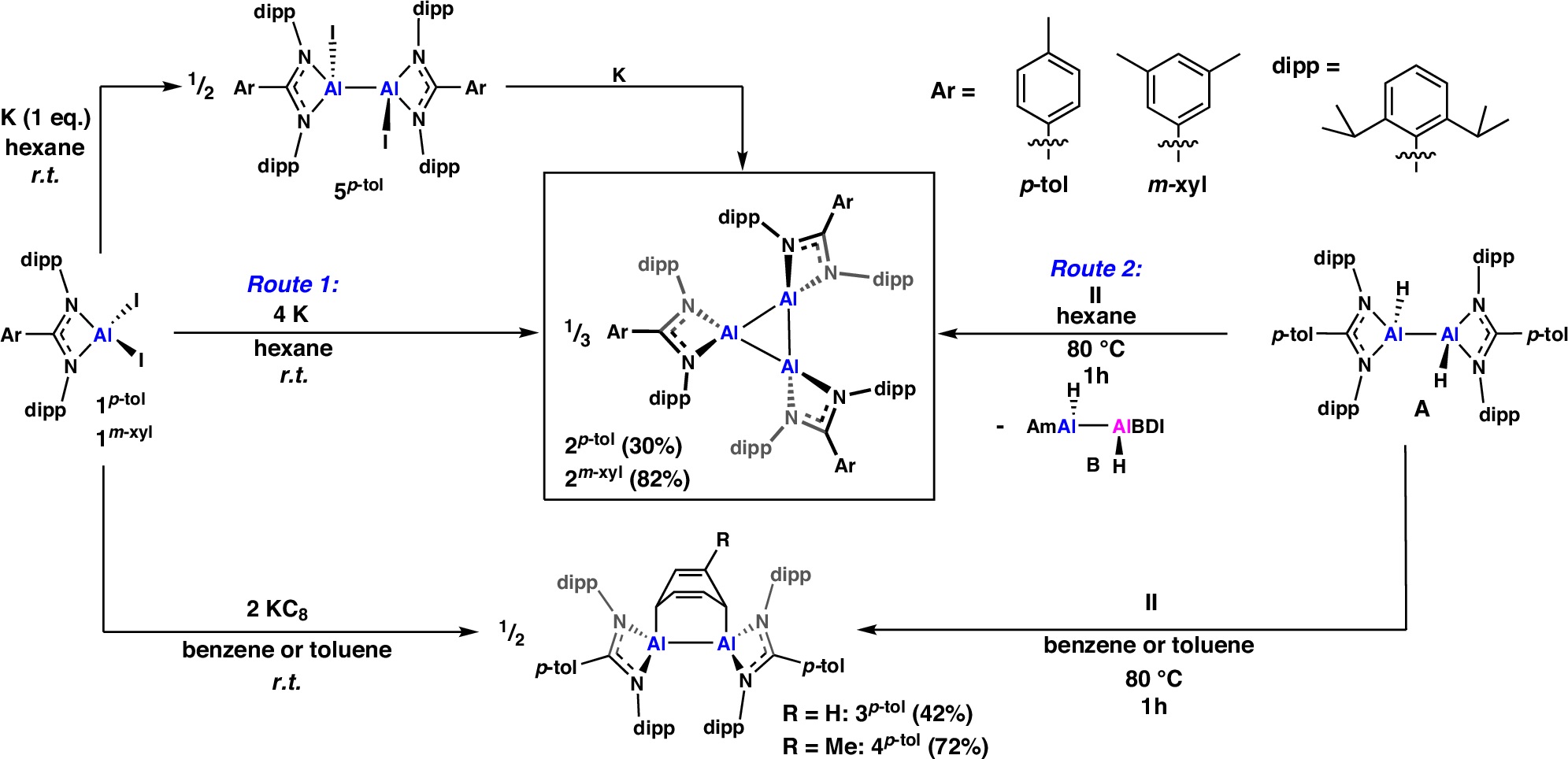
Synthetic routes to the formation of compounds 2p-tol and 2m-xyl (via routes 1 and 2) and the related products 3p-tol, 4p-tol and 5p-tol. Reaction conditions, times and yields shown in the figure.
image source: nature.com
The research team, led by Dr. Clare Bakewell from the Department of Chemistry at King’s College London, created highly reactive aluminum-based molecules capable of breaking some of the strongest chemical bonds known in chemistry.
One of the most significant achievements of the study was the creation of the first known cyclotrialumane.
This unusual compound consists of three aluminum atoms linked together in a triangular structure, forming what scientists call an aluminum trimer.
Despite its high reactivity, the molecule remains stable when dissolved in different chemical environments, which allows it to participate in a wide variety of reactions.
This balance between reactivity and stability makes it particularly valuable for future chemical applications.
advertisement
Unlocking New Chemical Reactions
The aluminum trimer has demonstrated the ability to perform chemical transformations typically associated with expensive transition metals.
Among its capabilities:
• Splitting dihydrogen molecules
• Controlling the insertion of ethene molecules
• Supporting chain growth reactions
Ethene, a simple two-carbon hydrocarbon, is a key building block in industrial chemical manufacturing, meaning this discovery could have significant implications for producing plastics, fuels, and other materials.
Even more remarkably, the aluminum compound can form new molecular structures, including five- and seven-membered rings made of aluminum and carbon — structures that had never been observed before.
These discoveries reveal entirely new pathways of chemical reactivity, expanding what scientists previously believed aluminum could do.
Reducing Dependence on Rare and Precious Metals
Many industrial chemical processes rely heavily on precious transition metals, such as platinum and palladium, because they act as highly effective catalysts.
However, these metals present several challenges:
• They are rare and expensive
• Mining them causes environmental damage
• Supply chains are often located in politically unstable regions
According to Dr. Bakewell, aluminum offers a promising alternative.
She explains that aluminum is approximately 20,000 times less expensive than precious metals like platinum and palladium, making it an attractive option for future chemical technologies.
By harnessing aluminum’s newly discovered properties, scientists could potentially create cost-effective catalysts and industrial processes that rely on far more abundant resources.
advertisement
Pushing the Boundaries of Chemical Science
While aluminum compounds have been studied for decades, this research pushes the element into entirely new territory.
The aluminum trimer developed in the study can produce new compounds with unprecedented levels of reactivity, potentially enabling chemists to design reactions that were previously impossible.
Dr. Bakewell emphasizes that the research is still in an early exploratory phase, but the potential applications are enormous.
Future research could lead to:
• New classes of chemical catalysts
• Advanced materials with unique molecular properties
• Cleaner and more efficient industrial chemical processes
advertisement
A Step Toward Greener and More Affordable Chemistry
If further research confirms the potential of this discovery, aluminum could become a key building block for sustainable chemistry.
Because it is abundant, inexpensive, and widely available, aluminum-based catalysts could significantly reduce the environmental and economic costs of producing chemicals, fuels, and advanced materials.
The discovery also highlights how rethinking familiar elements can lead to revolutionary innovations.
Instead of relying on rare resources, scientists may increasingly turn to earth-abundant materials to power the technologies of the future.
Conclusion
The discovery of a new highly reactive aluminum trimer marks an exciting milestone in modern chemistry.
By demonstrating that aluminum can perform reactions previously dominated by rare and precious metals, researchers have opened the door to more sustainable, affordable, and environmentally friendly chemical production.
Although the research is still in its early stages, the possibilities are vast. From innovative catalysts to entirely new materials, this breakthrough could reshape industries that depend on complex chemical processes.
In the long run, this discovery reminds us that some of the most powerful scientific revolutions begin not with rare elements, but with reimagining the potential of the materials we already have in abundance.
Key Points
• Scientists discovered a new triangular aluminum molecule called cyclotrialumane.
• The compound can perform powerful chemical reactions previously associated with precious metals.
• Aluminum is far cheaper and more abundant, making it a promising alternative to rare earth metals.
• The breakthrough could enable greener and more sustainable industrial chemistry.
• Researchers believe this discovery may lead to new materials and catalytic technologies.
advertisement
Frequently Asked Questions (FAQ)
1- What is the new form of aluminum discovered by scientists?
Researchers created a molecule called cyclotrialumane, which consists of three aluminum atoms connected in a triangular structure.
2- Why is this discovery important?
The aluminum compound can perform chemical reactions typically carried out by rare and expensive metals, potentially reducing costs and environmental impact.
3- Can aluminum really replace rare earth metals?
It may not replace them in every application yet, but the discovery shows that aluminum could perform similar catalytic roles in some chemical processes.
4- Why are scientists looking for alternatives to rare metals?
Rare metals are expensive, environmentally costly to mine, and often difficult to obtain, making sustainable alternatives highly desirable.
5- Is this technology ready for industry?
Not yet. Researchers say the work is still in the exploratory stage, but the early results are promising.
Sources
- SciTechDaily – Report on the discovery of a new aluminum structure that could replace rare earth metals
https://scitechdaily.com/scientists-create-powerful-new-form-of-aluminum-that-could-replace-rare-earth-metals/ - Nature Communications – Scientific paper describing the neutral cyclic aluminum (I) trimer
https://www.nature.com/articles/s41467-026-68432-1
Thank you !